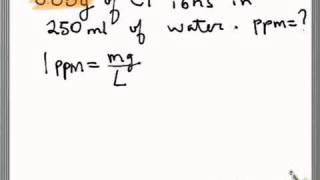![O3 concentration [ppm] in oxygen at various flows ranging from 1.9 to... | Download Scientific Diagram O3 concentration [ppm] in oxygen at various flows ranging from 1.9 to... | Download Scientific Diagram](https://www.researchgate.net/publication/332716357/figure/fig11/AS:962137216266241@1606402796212/O3-concentration-ppm-in-oxygen-at-various-flows-ranging-from-19-to-10-slm.png)
O3 concentration [ppm] in oxygen at various flows ranging from 1.9 to... | Download Scientific Diagram

A sample of 300.0 g of drinking water is found to contain 38 mg Pb. What this concentration in parts per million?a)3 x 102ppmb)6.5 mc)130 ppm Pbd)21 ppmCorrect answer is 'C'. Can
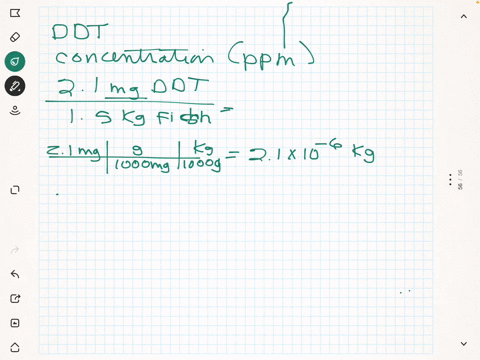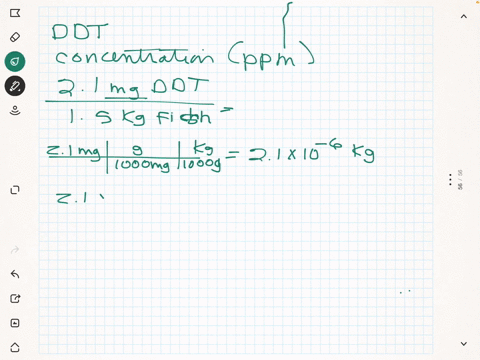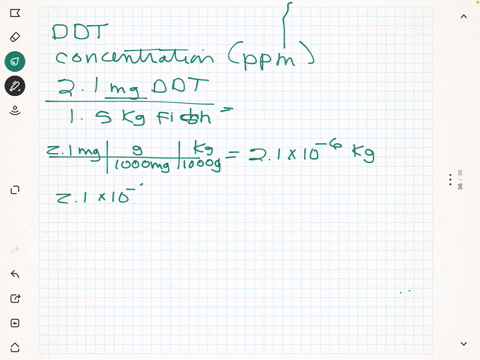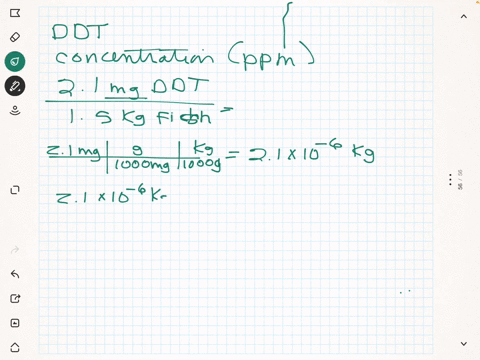
SOLVED: What is the concentration in parts per million of DDT in each of the following 0.037 mg in 1200 g plankton Ppm DDT b.2.7 mg in L8 kg needlefish tissue Ppm DDT

SOLVED: What is the ppm concentration of 6.00 mL sample of solution that has 3.6 x 10-4 g of sodium ions? 20 60 40 80

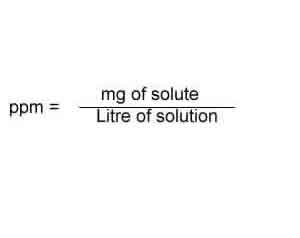
SOLVED: In clinical applications, the unit parts per million (ppm) is used to express very small concentrations of solute, where ppm equivalent to mg of solute per 1 L of solution. Calculate

Parts per Million Concentration. Parts per Million (ppm) is the ratio of the number of grams of solute for every one million grams of solution. parts. - ppt download

To find out the concentration of SO2 in the air (in parts per million, i.e., ppm), the data was collected for 30 localities in a certain city and is presented below: Find