Commission Delegated Regulation (EU) 2019/2017 of 11 March 2019 supplementing Regulation (EU) 2017/1369 of the European Parliame
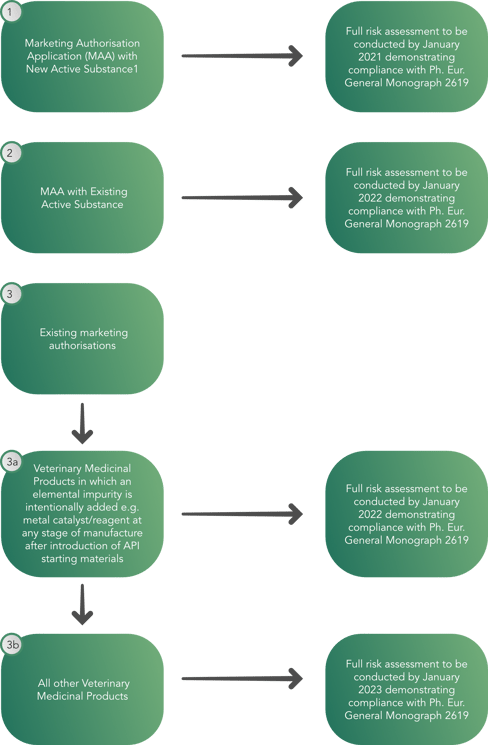
COMMISSION DELEGATED REGULATION (EU) 2021/577 of 29 January 2021 supplementing Regulation (EU) 2019/6 of the European Parliament
6908/23 OT/mb LIFE.3 Delegations will find attached document C(2023) 1272 final. Encl.: C(2023) 1272 final

Italy | Regulation (EU) 2019/6: Implementing acts on good pharmacovigilance practice for veterinary medicinal products and on the format and content of the reference dossier of the pharmacovigilance system