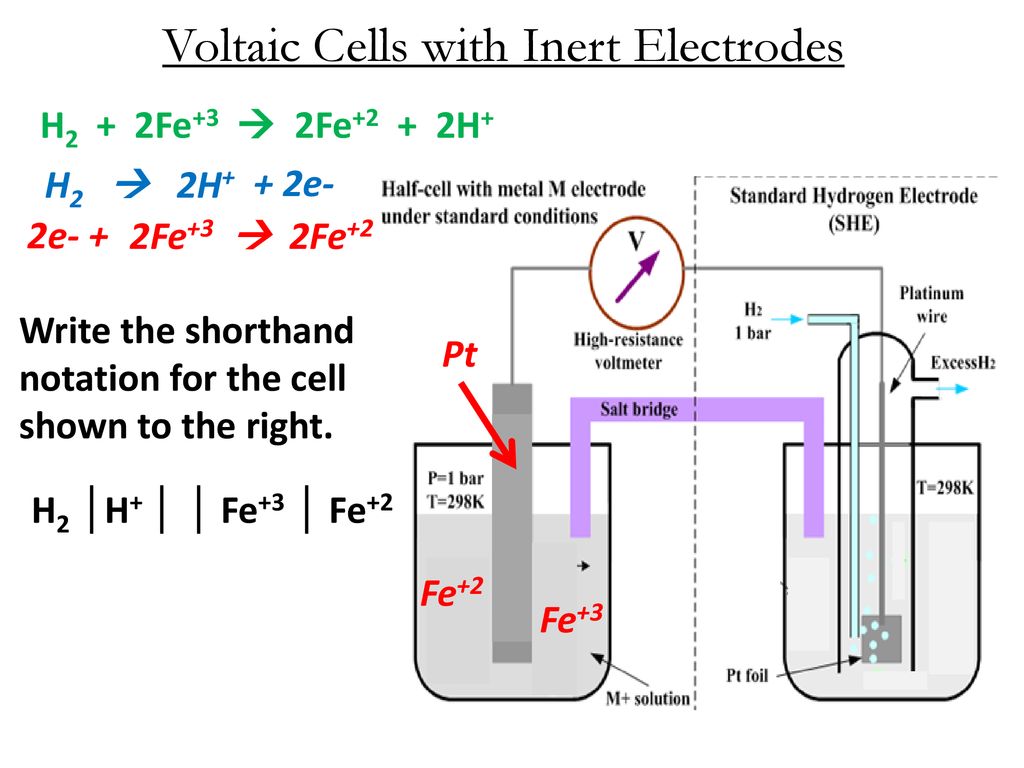
Electrode Materials in Modern Organic Electrochemistry - Heard - 2020 - Angewandte Chemie International Edition - Wiley Online Library

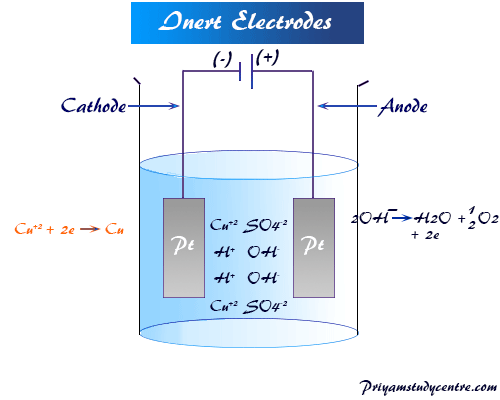
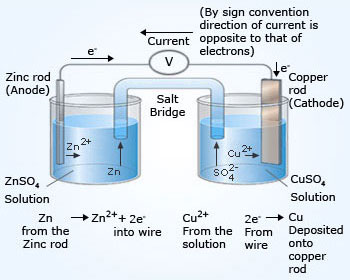
electrolysis copper sulfate solution with copper carbon graphite electrodes electroplating half-equations products anode cathode apparatus electrolyte cell anode sludge gcse chemistry KS4 science igcse O level revision notes
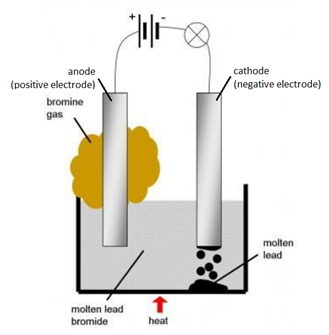
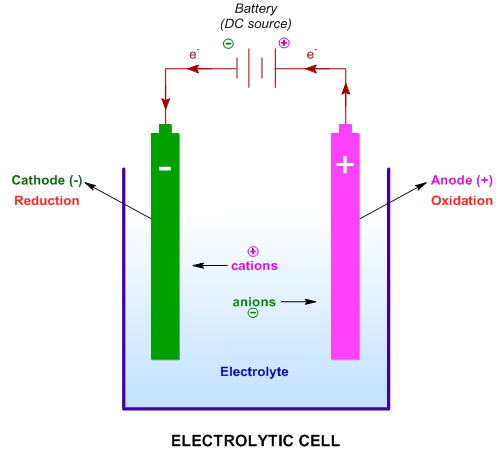
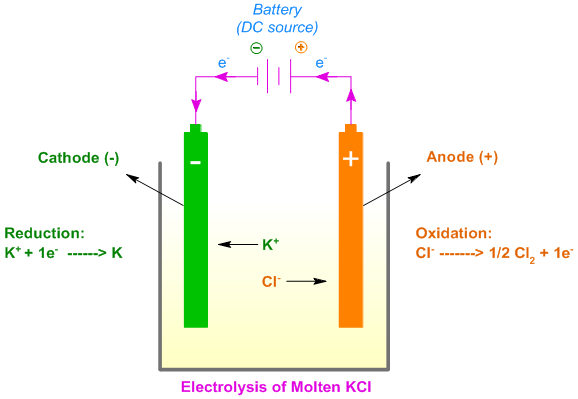
1:58 (Triple only) describe experiments to investigate electrolysis, using inert electrodes, of molten compounds (including lead(II) bromide) and aqueous solutions (including sodium chloride, dilute sulfuric acid and copper(II) sulfate) and to predict