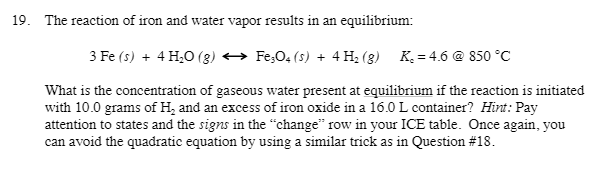Class 9 Chemistry Practical's and Experiments on To carry out the reaction of Iron with copper sulphate solution in water and classify it as physical or chemical changes

Substituting Chromium in Iron-Based Catalysts for the High-Temperature Water–Gas Shift Reaction | ACS Catalysis

Write equations for the reactions of (i) iron with steam (ii) calcium and potassium with water... - YouTube

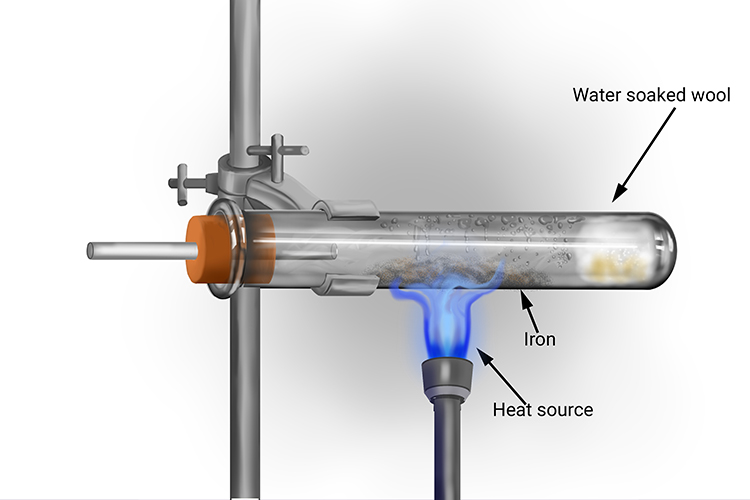
The reaction of iron and water vapor results in an equilibrium reaction. 3Fe(s) + 4H_2O(g) rightarrow Fe_3O_4(s) + 4H_2(g) An equilibrium constant of 4.6 at 850 degree C. What is the concentration

Chemical equation for the following reaction. Phases are optional. Solid iron(III) oxide reacts with hydrogen - Home Work Help - Learn CBSE Forum
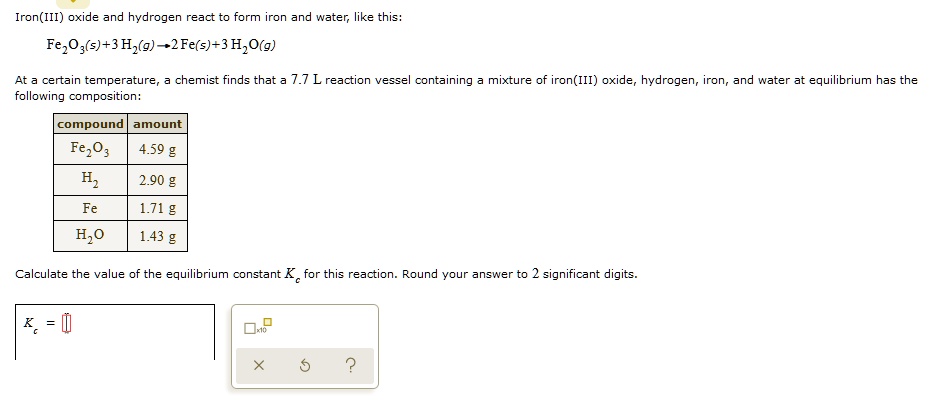
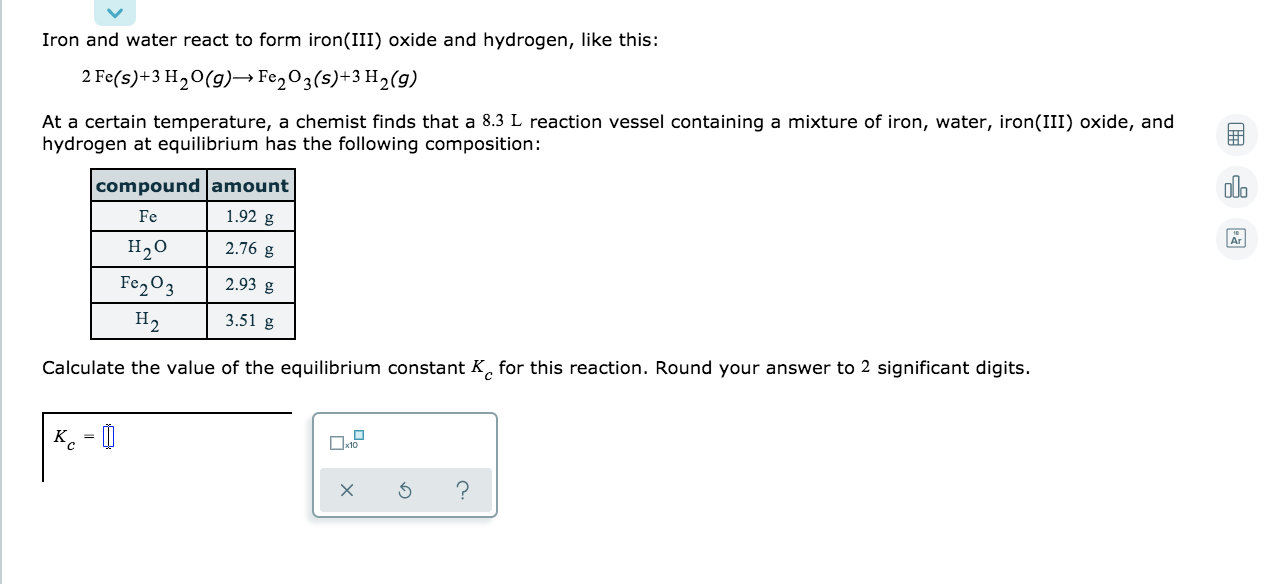
SOLVED: Iron(III) oxide and hydrogen react to form iron and water like this= FezO3(s)+3H,(g)-2Fe(s)-3 H,O(g) At a certain temperature chemist finds that 7.7 L reaction vessel containing mixture of iron(III) oxide; hydrogen;

Iron will react with water to produce an iron oxide and hydrogen gas. Which equation below represents a - Brainly.com

Arrange the following metal in the increasing order of their reactivity towards water: Zinc, Iron, Magnesium, Sodium.

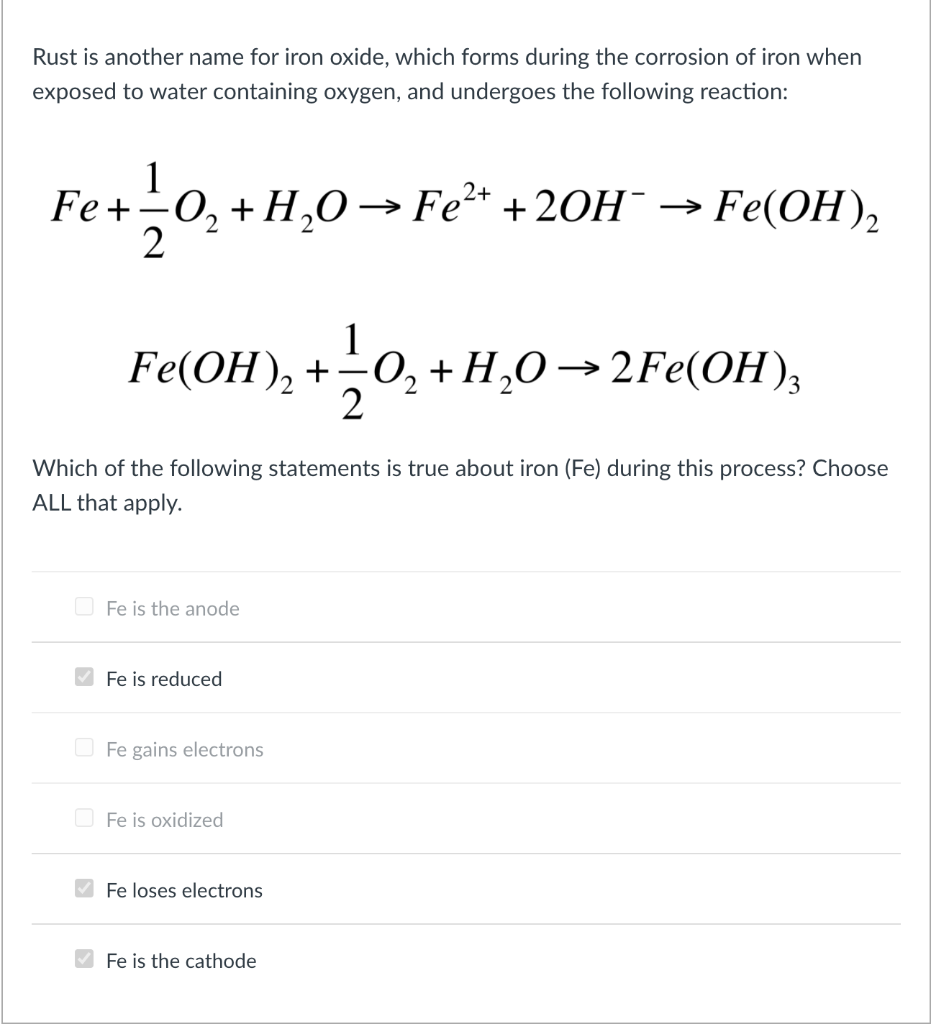
Iron + Water = Rust | Chemistry! | Balancing Equations, Coefficients, Molecules - Science Q&A - YouTube