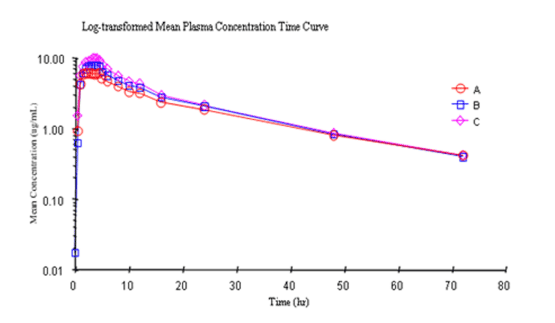
Calibration plot of log current Vs log concentration of substrate ATCl... | Download Scientific Diagram

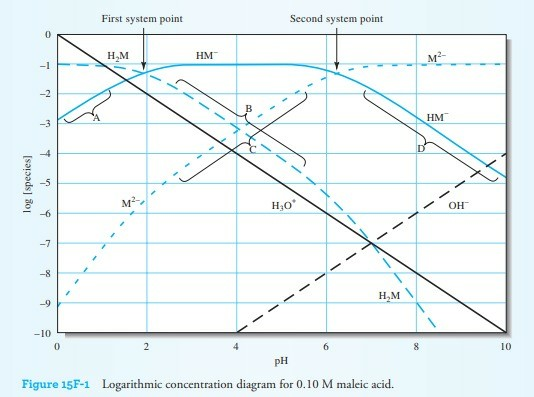
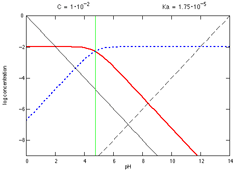
Using the Logarithmic Concentration Diagram, Log C, To Teach Acid–Base Equilibrium | Journal of Chemical Education

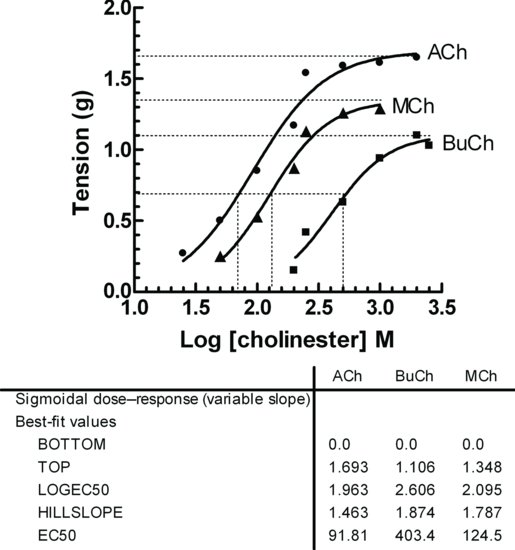
Dose Response Curve | Effect, Equation, Pharmacology & Examples - Video & Lesson Transcript | Study.com

A graph between log t1/2 and log a (abscissa), 'a' is the initial concentration of A in the reaction, is given. For the reaction, A → Product, the rate law is:
![Following is the graph between log ${{T}_{{1}\/{2}\\;}}$ and log a (a = initial concentration) for a given reaction at ${{27}^{{}^\\circ }}C$ . Hence, the order is:\n \n \n \n \n [A] 0[B] Following is the graph between log ${{T}_{{1}\/{2}\\;}}$ and log a (a = initial concentration) for a given reaction at ${{27}^{{}^\\circ }}C$ . Hence, the order is:\n \n \n \n \n [A] 0[B]](https://www.vedantu.com/question-sets/527f0926-d8c9-4fbf-9c13-90d60ec0e8696127764744518469031.png)
Following is the graph between log ${{T}_{{1}\/{2}\\;}}$ and log a (a = initial concentration) for a given reaction at ${{27}^{{}^\\circ }}C$ . Hence, the order is:\n \n \n \n \n [A] 0[B]
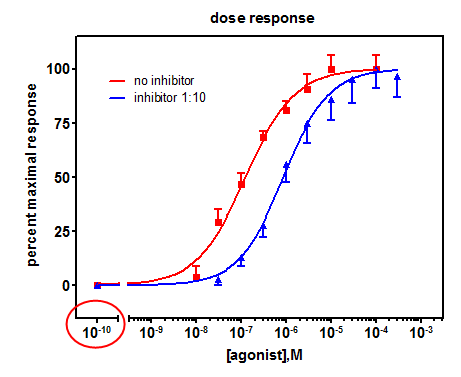
How can I put a zero concentration point on my semi-log plot (e.g., competitive binding curve or dose-response curve)? - FAQ 165 - GraphPad

A plot of logarithm of rate vs logarithm of concentration of the reactant in a first order reaction - YouTube
![SOLVED: You graph the natural log of concentration (In [A]) vs. time from a table of data. You don't get a line. What does this mean? How can you determine (using a SOLVED: You graph the natural log of concentration (In [A]) vs. time from a table of data. You don't get a line. What does this mean? How can you determine (using a](https://cdn.numerade.com/ask_images/48fbdb48222a4c75af9cb9fd62747c68.jpg)
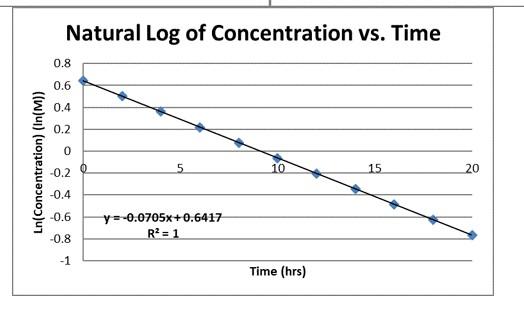
SOLVED: You graph the natural log of concentration (In [A]) vs. time from a table of data. You don't get a line. What does this mean? How can you determine (using a