
A STUDY OF PROCEDURES FOR DOSSIER PREPARATION AND THEIR MARKETING AUTHORISATION IN DIFFERENT COUNTRIES OF SELECTED DRUG(S) | PharmaTutor
![Amazon.in: Buy Comprehensive Technical Dossier (CTD) of Medicinal Plant Used in Ayurveda: Aksoda (Juglans Regia L.) [Series-I Number-4] Book Online at Low Prices in India | Comprehensive Technical Dossier (CTD) of Medicinal Amazon.in: Buy Comprehensive Technical Dossier (CTD) of Medicinal Plant Used in Ayurveda: Aksoda (Juglans Regia L.) [Series-I Number-4] Book Online at Low Prices in India | Comprehensive Technical Dossier (CTD) of Medicinal](https://m.media-amazon.com/images/I/A1ZANCZvmaL.jpg)
Amazon.in: Buy Comprehensive Technical Dossier (CTD) of Medicinal Plant Used in Ayurveda: Aksoda (Juglans Regia L.) [Series-I Number-4] Book Online at Low Prices in India | Comprehensive Technical Dossier (CTD) of Medicinal

Regulatory requirement for the approval of Generic Drug in Cambodia as per ASEAN Common Technical Dossier (ACTD) | Semantic Scholar





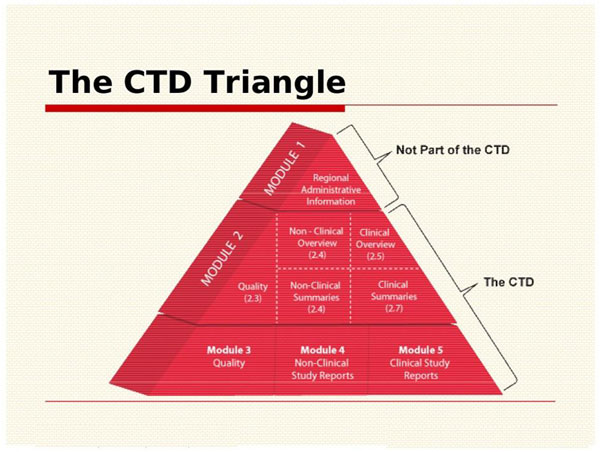









![Common technical document triangle [7] | Download Scientific Diagram Common technical document triangle [7] | Download Scientific Diagram](https://www.researchgate.net/publication/320372658/figure/fig1/AS:550839028203520@1508341666076/Common-technical-document-triangle-7.png)

