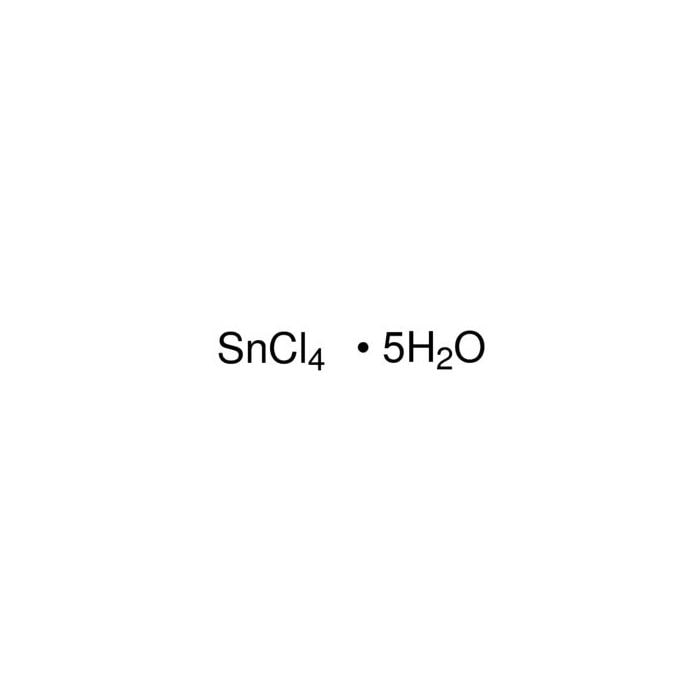
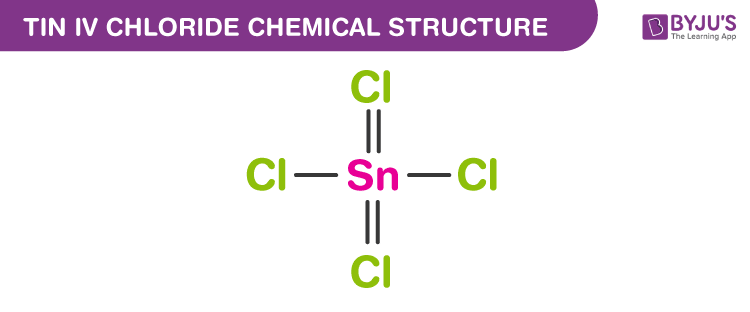
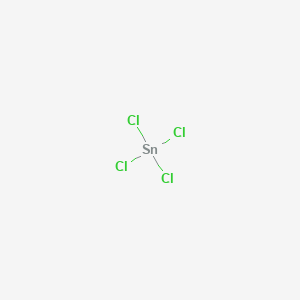
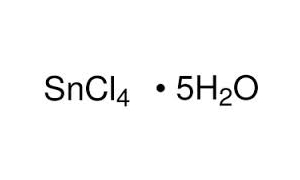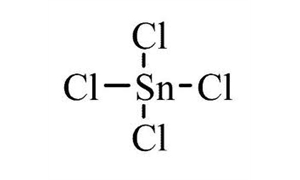Write the formula of the following compunds: (i)Tin (IV) oxide ,(ii) Copper (I) iodide ,(iii) chromium (III) oxide ,(IV) Thallium (I) suplhate ,(V) Bismuth (III) niitrate Mercury (II) chloride

SOLVED: Write the chemical formula for the following compounds Iron (II) sulfide b) Sodium nitrate c) Tin (IV) chloride d) Potassium sulfate Calcium hydroxide f) Copper (II) sulfate Magnesium nitrite

Naming Compounds PSc Naming Ionic Binary Compounds Metal -- Nonmetal NaClSodium Chloride KBrPotassium Bromide Calcium Oxide CaO First element – - ppt download