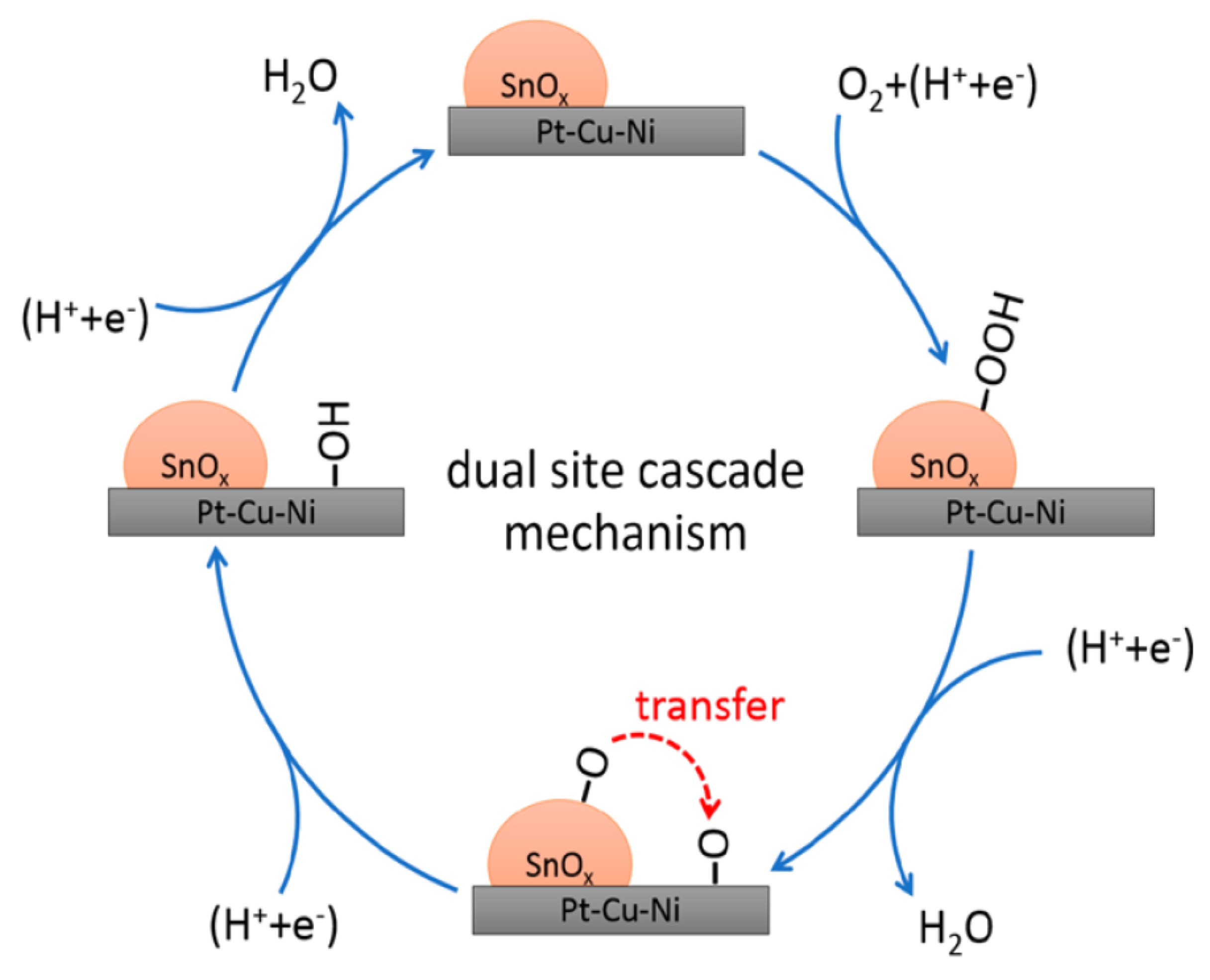
Tin Oxide Dependence of the CO2 Reduction Efficiency on Tin Electrodes and Enhanced Activity for Tin/Tin Oxide Thin-Film Catalysts | Journal of the American Chemical Society
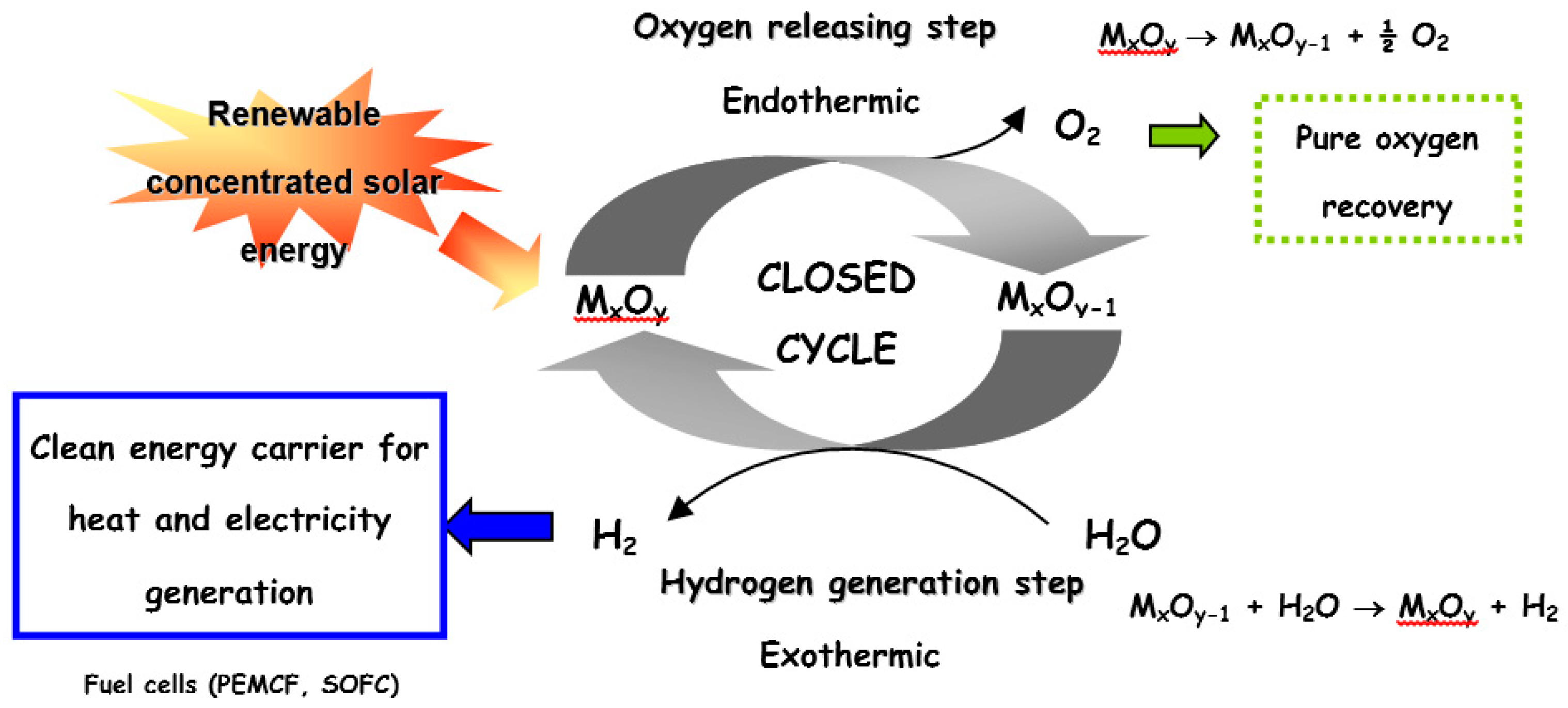
ChemEngineering | Free Full-Text | Metal Oxides Applied to Thermochemical Water-Splitting for Hydrogen Production Using Concentrated Solar Energy

Green and Cost-Effective Synthesis of Tin Oxide Nanoparticles: A Review on the Synthesis Methodologies, Mechanism of Formation, and Their Potential Applications | SpringerLink

Tin Oxide Dependence of the CO2 Reduction Efficiency on Tin Electrodes and Enhanced Activity for Tin/Tin Oxide Thin-Film Catalysts | Journal of the American Chemical Society

Reaction Mechanisms of the Electrochemical Conversion of Carbon Dioxide to Formic Acid on Tin Oxide Electrodes - Lee - 2017 - ChemElectroChem - Wiley Online Library

Green Biosynthesis of Tin Oxide Nanomaterials Mediated by Agro-Waste Cotton Boll Peel Extracts for the Remediation of Environmental Pollutant Dyes | ACS Omega

SOLVED: The reduction of tin (IV) oxide via carbon monoxide will produce tin metal and carbon dioxide tnrongh following reaction at 25"C: OVERALL SnOzt) CO,() The stepwise reaction and their 'corresponding equilibrium

Mechanism for the formation of tin oxide nanoparticles and nanowires inside the mesopores of SBA-15 - ScienceDirect
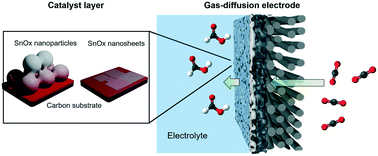
Unveiling the effects of dimensionality of tin oxide-derived catalysts on CO2 reduction by using gas-diffusion electrodes - Reaction Chemistry & Engineering (RSC Publishing)
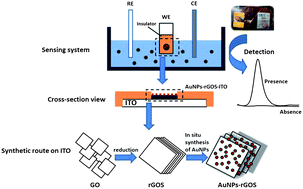
Indium tin oxide-coated glass modified with reduced graphene oxide sheets and gold nanoparticles as disposable working electrodes for dopamine sensing in meat samples - Nanoscale (RSC Publishing)

Superior performance of rGO-tin oxide nanocomposite for selective reduction of CO2 to methanol - ScienceDirect

Tin Oxide Based Nanomaterials and Their Application as Anodes in Lithium‐Ion Batteries and Beyond - Zoller - 2019 - ChemSusChem - Wiley Online Library
![PDF] Tin nanoparticles decorated copper oxide nanowires for selective electrochemical reduction of aqueous CO2 to CO | Semantic Scholar PDF] Tin nanoparticles decorated copper oxide nanowires for selective electrochemical reduction of aqueous CO2 to CO | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/5089acbf8f19b13171cbc7d76afbef6aff6000f9/11-Figure1-1.png)
PDF] Tin nanoparticles decorated copper oxide nanowires for selective electrochemical reduction of aqueous CO2 to CO | Semantic Scholar

Tin Oxide Dependence of the CO2 Reduction Efficiency on Tin Electrodes and Enhanced Activity for Tin/Tin Oxide Thin-Film Catalysts | Journal of the American Chemical Society

Reduction behavior of SnO2 in the tin-bearing iron concentrates under CO–CO2 atmosphere. Part II: Effect of quartz - ScienceDirect

Defect-engineering of tin oxide via (Cu, N) co-doping for electrocatalytic and photocatalytic CO2 reduction into formate - ScienceDirect