Bsi MD MDR Best Practice Documentation Submissions | PDF | Verification And Validation | Quality Management System
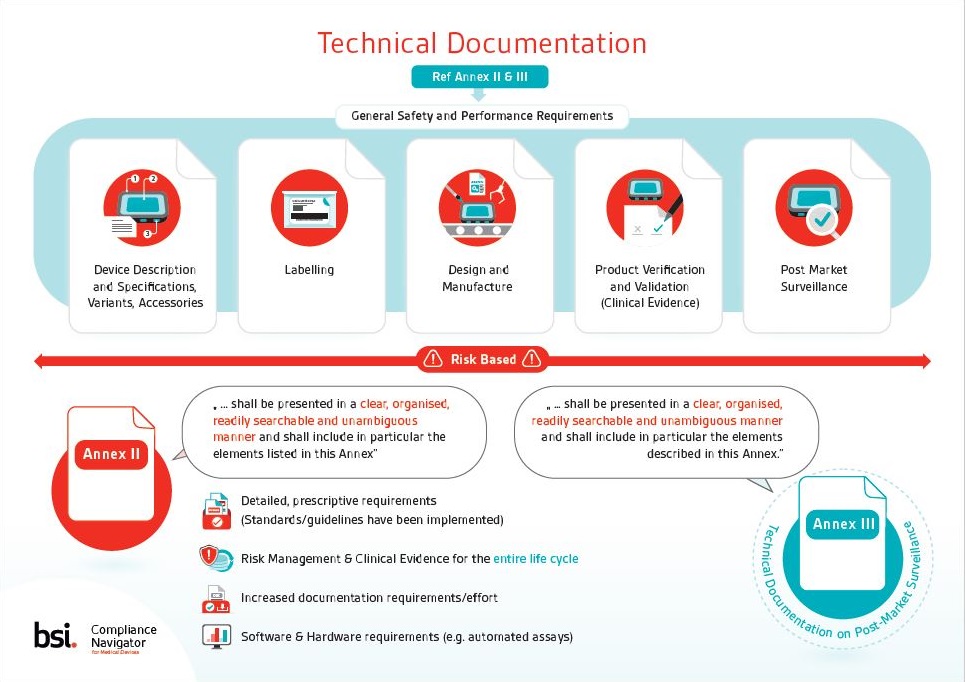
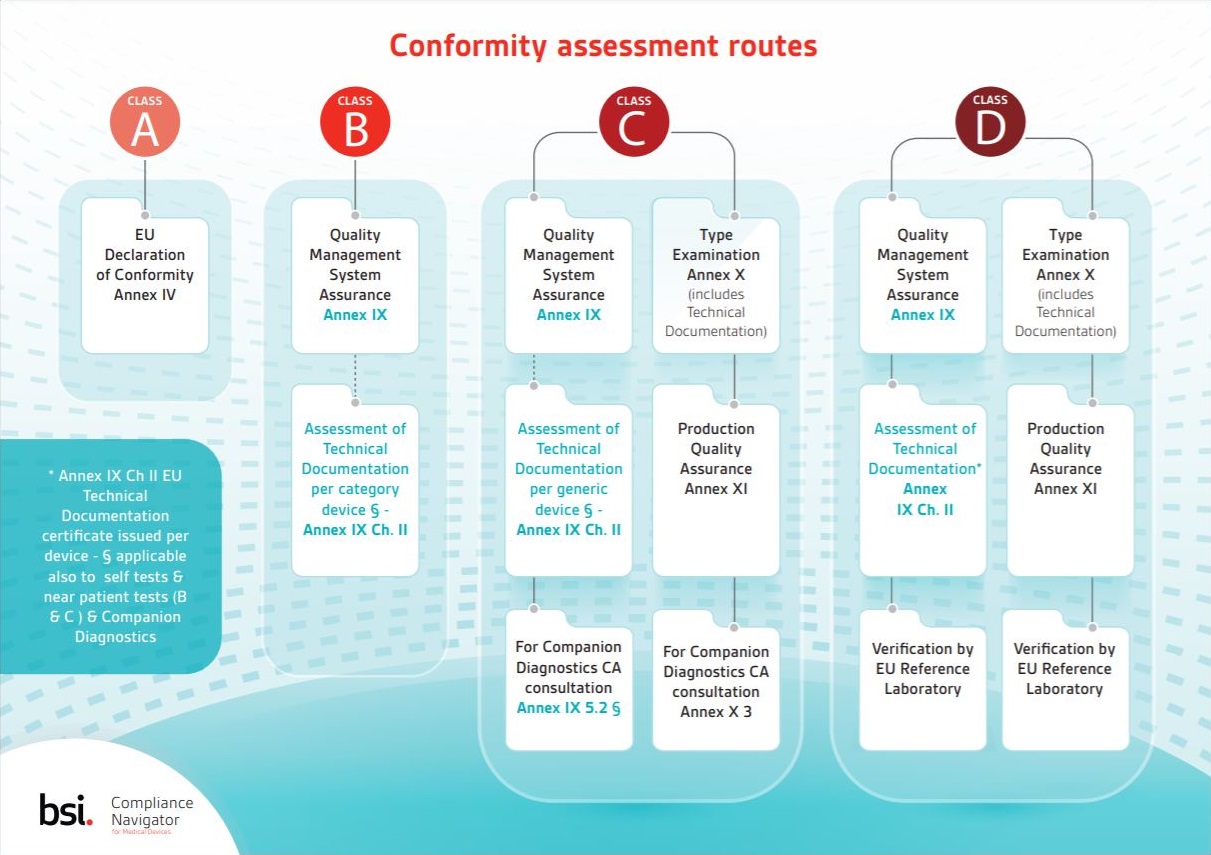
▫ Device Classification ▫ Conformity Assessment ▫ Safety & Performance Requirements ▫ Technical Documentation

Bsi MD MDR Best Practice Documentation Submissions en GB | PDF | Verification And Validation | Medical Device