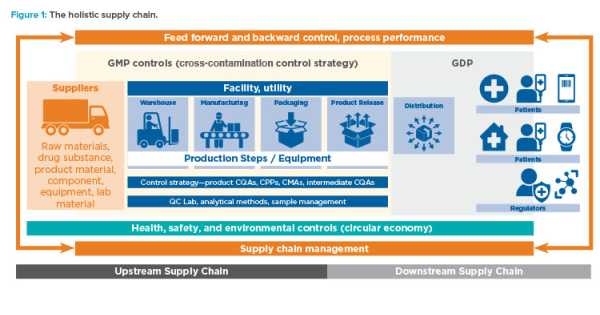
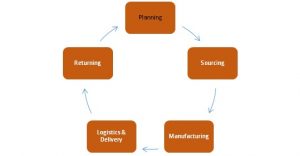
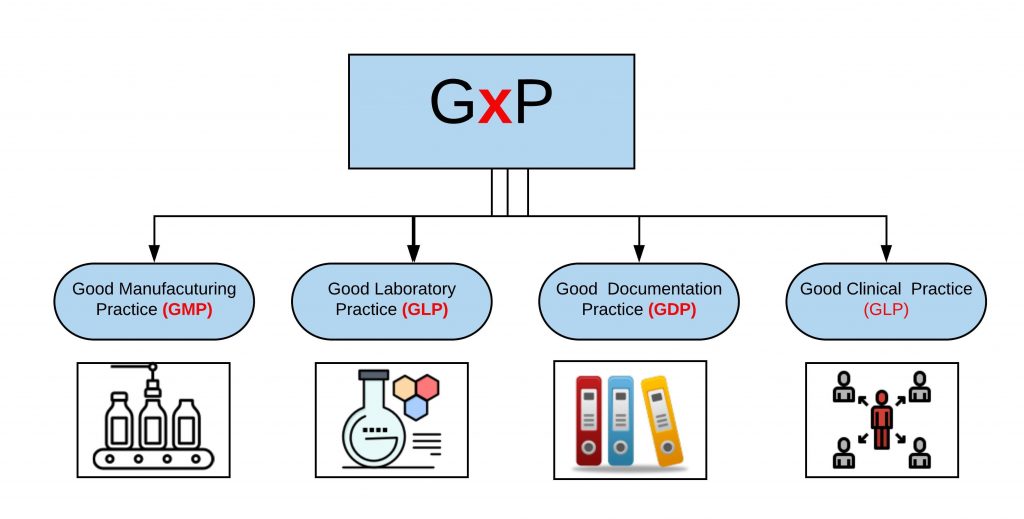
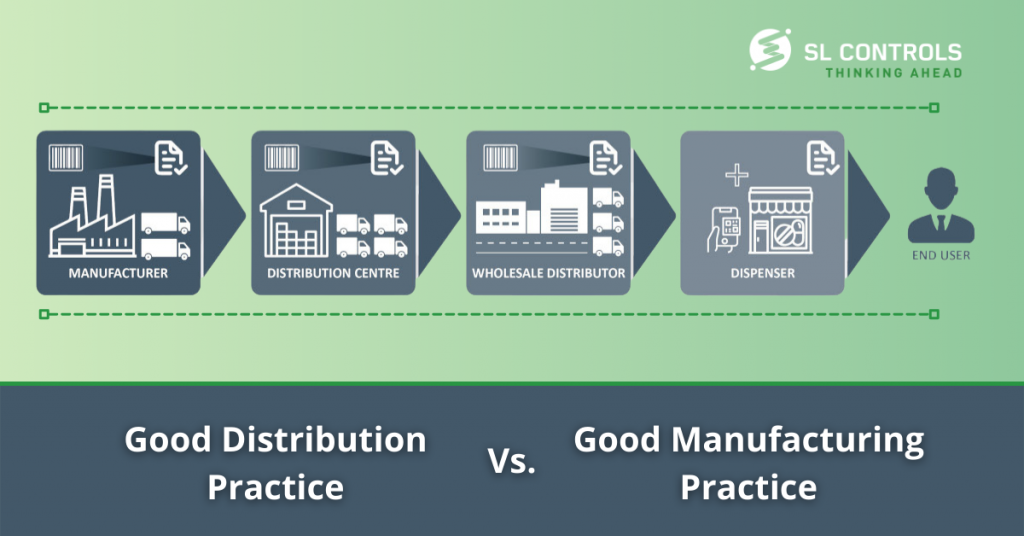
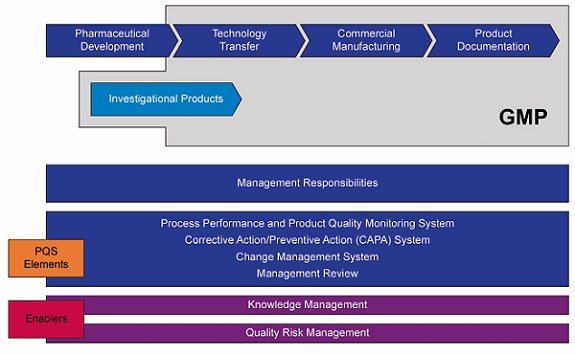
Mapping the Secure Temperature-Controlled Supply Chain Requirements | Pharmaceutical Outsourcing - The Journal of Pharmaceutical & Biopharmaceutical Contract Services
Template for the Qualified Person's Declaration Concerning GMP Compliance of the Active Substance Used as Starting Material an

GMP LOGFILE: Lead Article - GMP-Verlag: Is COVID-19 THE Long Overdue Wake-up Call for Pharma Supply-Chains?

A Green Chemistry Continuum for a Robust and Sustainable Active Pharmaceutical Ingredient Supply Chain | ACS Sustainable Chemistry & Engineering
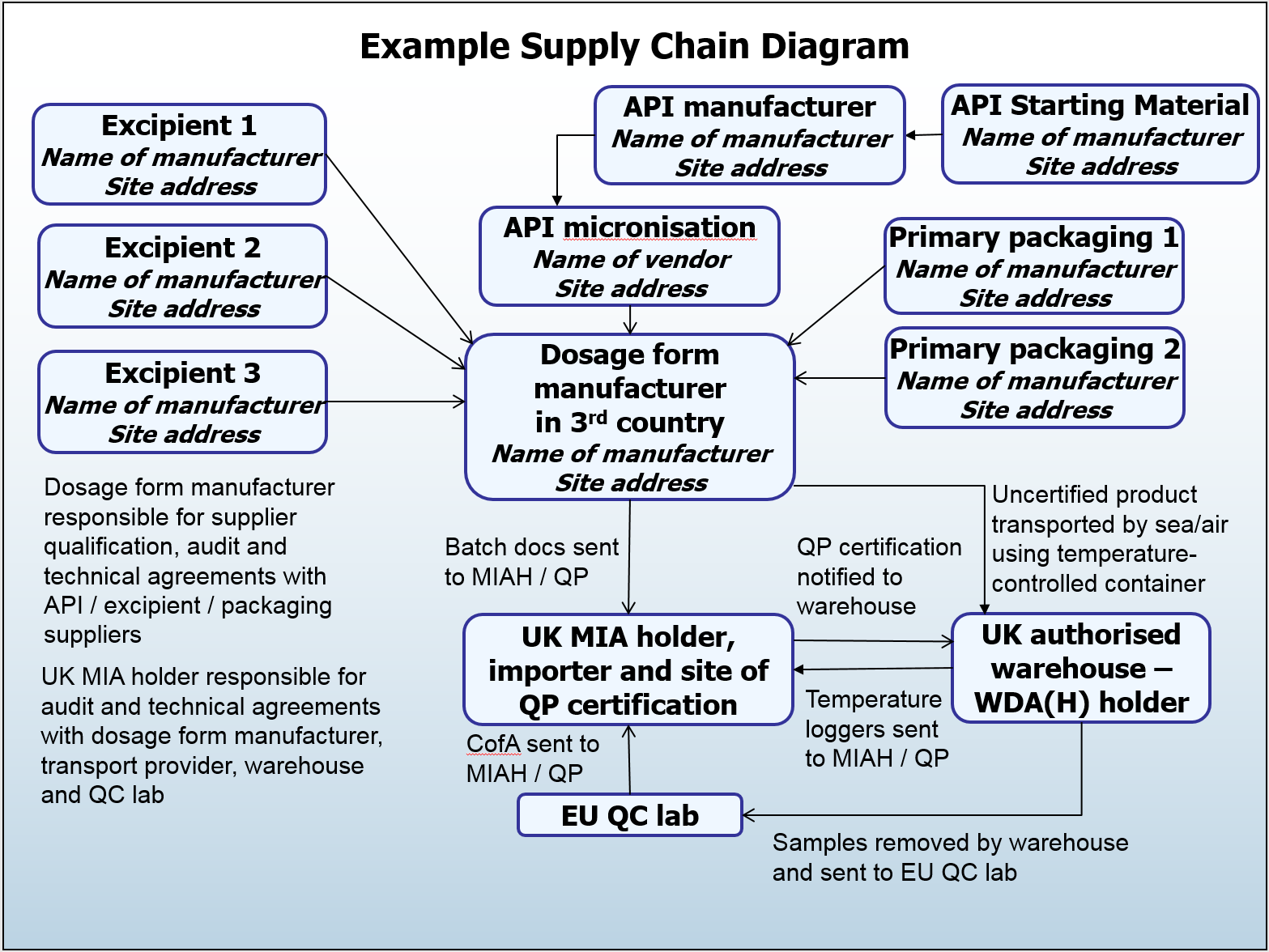
Annex 16 QP certification and batch release – frequently asked questions – part 1 - MHRA Inspectorate