SOLVED: Iron (III) iodide reacts with bromine to produce iodine and iron(III) bromide. 2 FeI3 + 3Br2 = 3I2 + 2FeBr a. If 218 g of iron (III) iodide reacts with 90.0

Profile of iodine liberation by oxidizing iodide ions with iron(III).... | Download Scientific Diagram

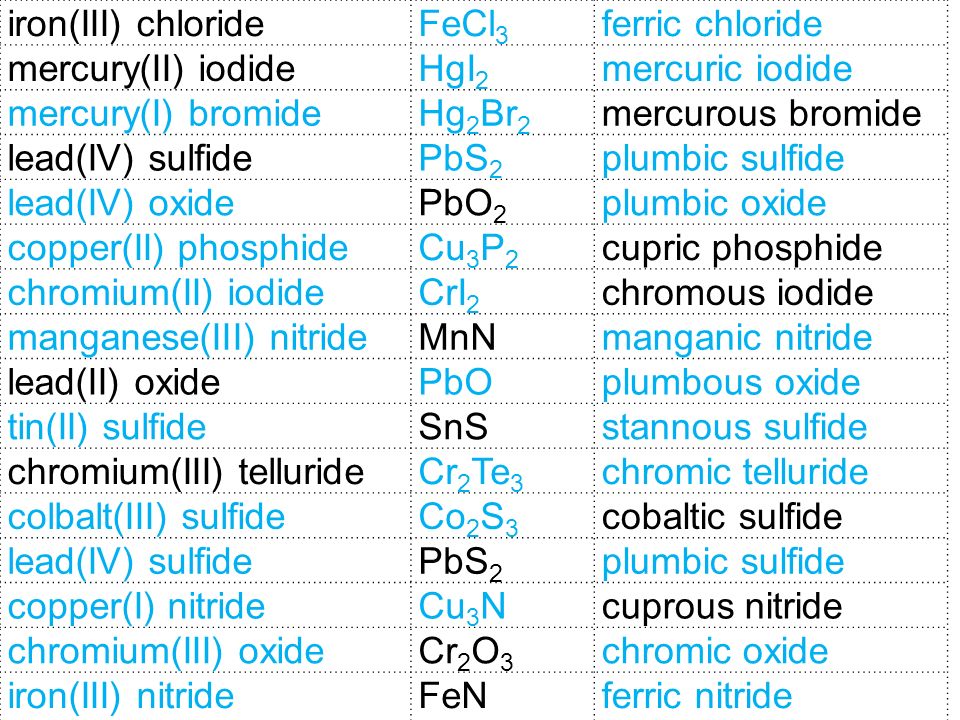
Iron transition metal Chemistry iron(II) Fe2+ iron(III) Fe3+ complexes ions ligand substitution redox chemical reactions principal oxidation states +2 +3 extraction GCE AS A2 IB A level inorganic chemistry revision notes
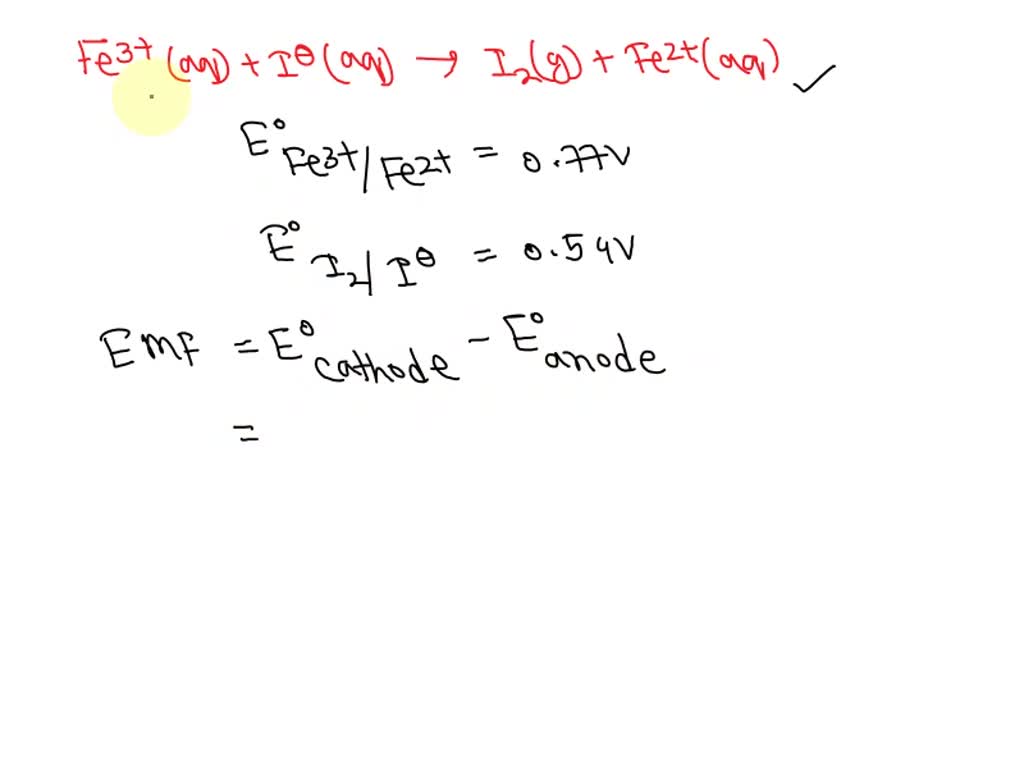
SOLVED: Would it be possible for iron(III) to oxidize iodide ion to iodine molecule under standard-state conditions? -No - It depends on the concentration of iron(III) and chloride ions. -Yes The calculated

Bismuth(III) iodide Magnesium iodide Bismuth chloride Crystal structure, iron, purple, electronics png | PNGEgg

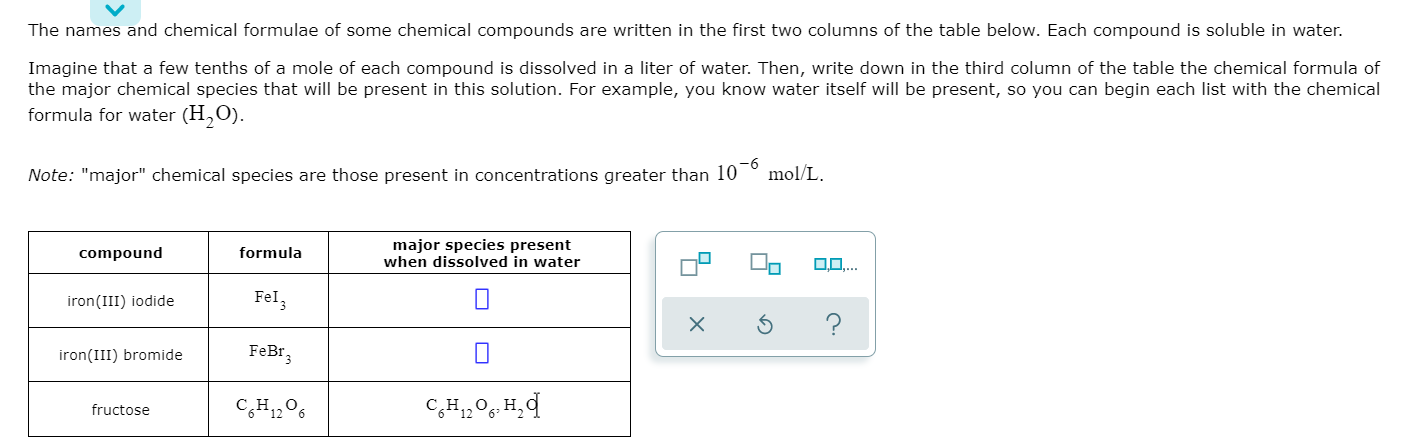
Solved! The names and chemical formulae of some chemical compounds are written in the first two columns of the table below. Each compound is soluble in water. Imagine that a few tenths
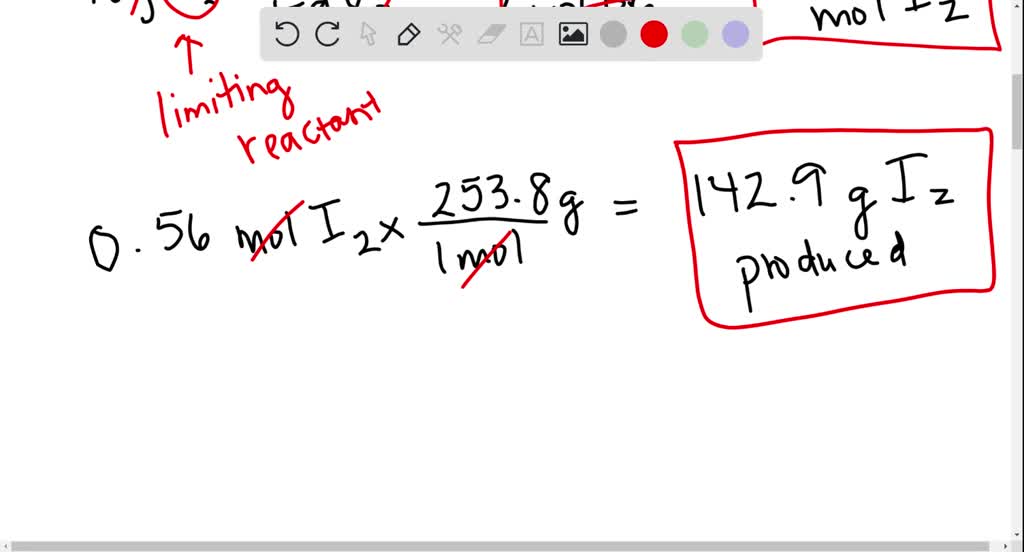
SOLVED: Iron (III) iodide reacts with bromine to produce iodine and iron(III) bromide. 2 FeI3 + 3Br2 = 3I2 + 2FeBr (theres no three in FeBr) a. If 218 g of iron (
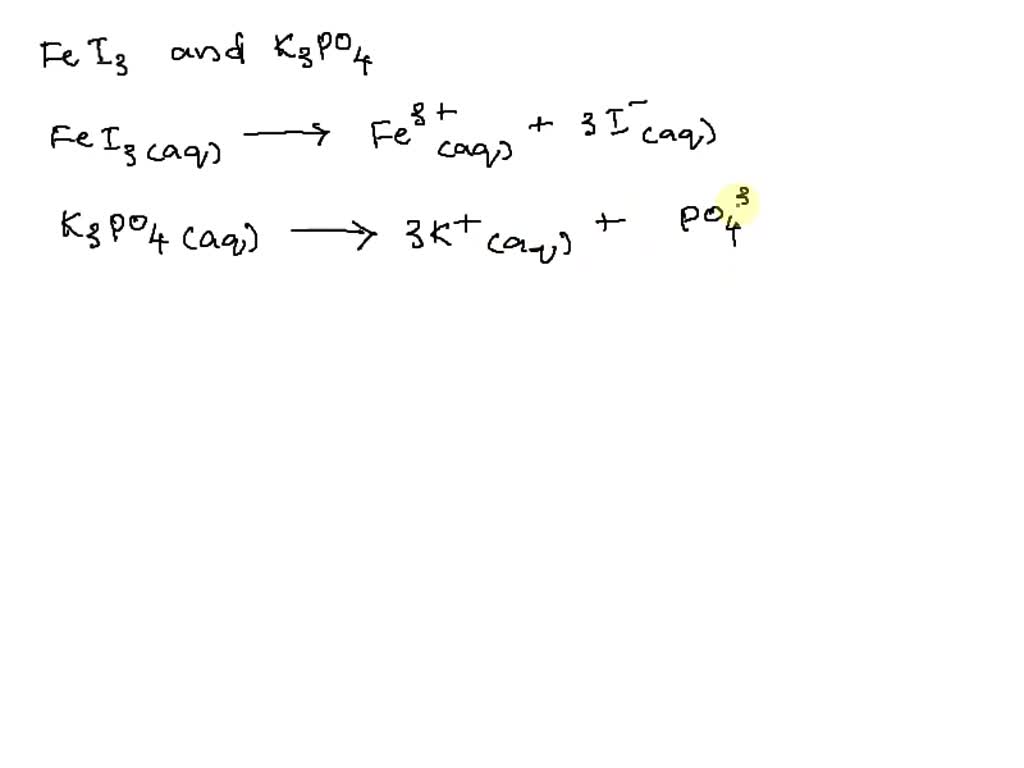


![ANSWERED] Iron (III) iodide reacts with bromine to... - Inorganic Chemistry ANSWERED] Iron (III) iodide reacts with bromine to... - Inorganic Chemistry](https://media.kunduz.com/media/sug-question/raw/49482159-1658689892.1666656.jpeg)