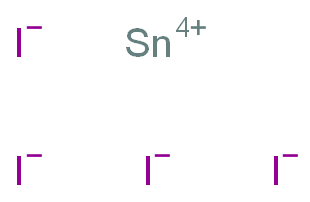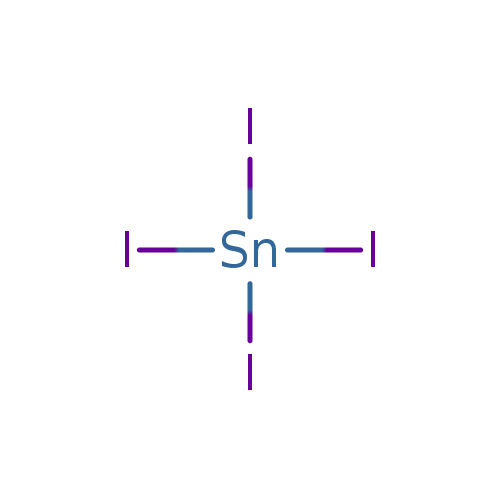Write the formula of the following compunds: (i)Tin (IV) oxide ,(ii) Copper (I) iodide ,(iii) chromium (III) oxide ,(IV) Thallium (I) suplhate ,(V) Bismuth (III) niitrate Mercury (II) chloride

Figure 1 from Influence of π-Iodide Intermolecular Interactions on Electronic Properties of Tin(IV) Iodide Semiconducting Complexes. | Semantic Scholar
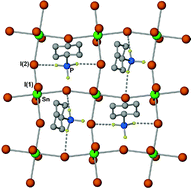
The reaction of tin(iv) iodide with phosphines: formation of new halotin anions - Dalton Transactions (RSC Publishing)
PDF) Influence of π-Iodide Intermolecular Interactions on Electronic Properties of Tin(IV) Iodide Semiconducting Complexes
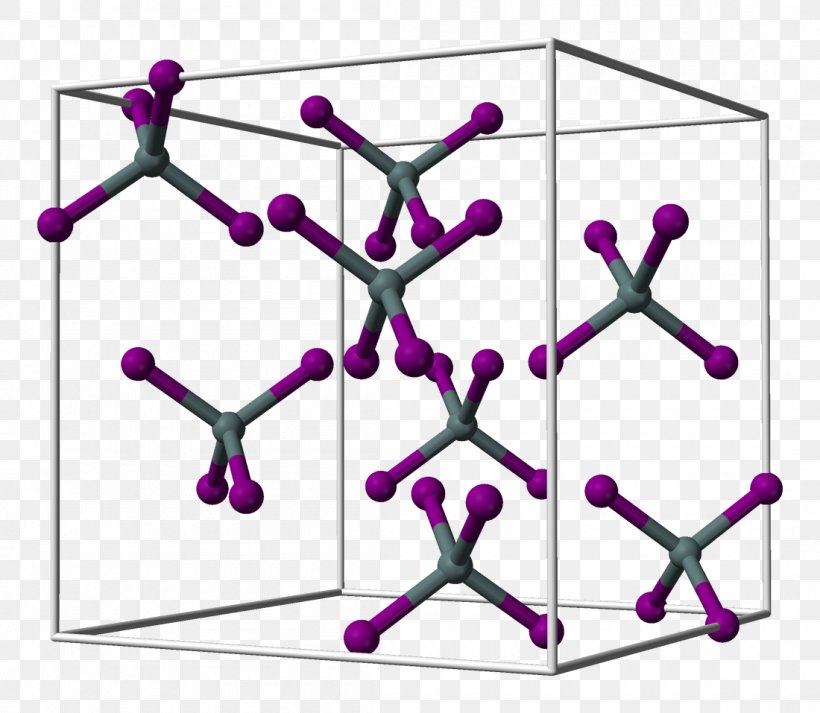
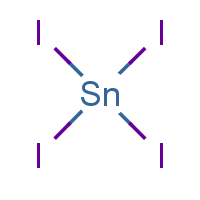
Tin(IV) Iodide Tin(IV) Chloride Crystal Structure, PNG, 1100x957px, Tiniv Iodide, Area, Body Jewelry, Carbon

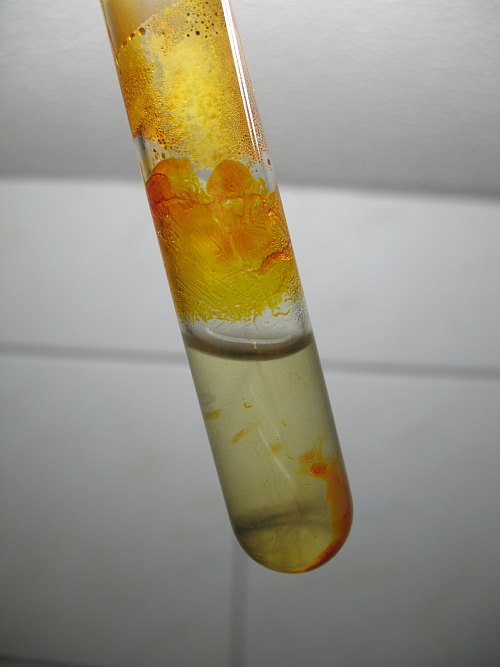
Tin IV Iodide is a covalent compound formed by directly combining tin metal with iodine. There should be a suitable solvent for that. I used Dichloromethane . The crystals of Tin IV

Figure 7 from Influence of π-Iodide Intermolecular Interactions on Electronic Properties of Tin(IV) Iodide Semiconducting Complexes. | Semantic Scholar