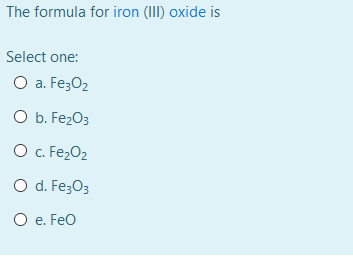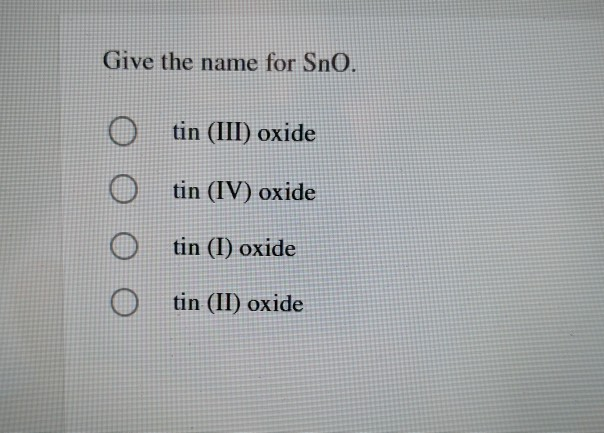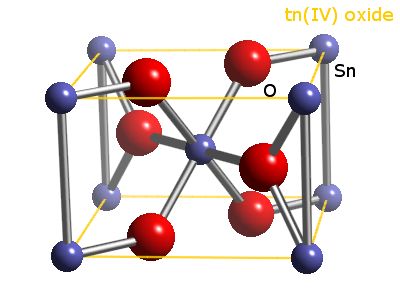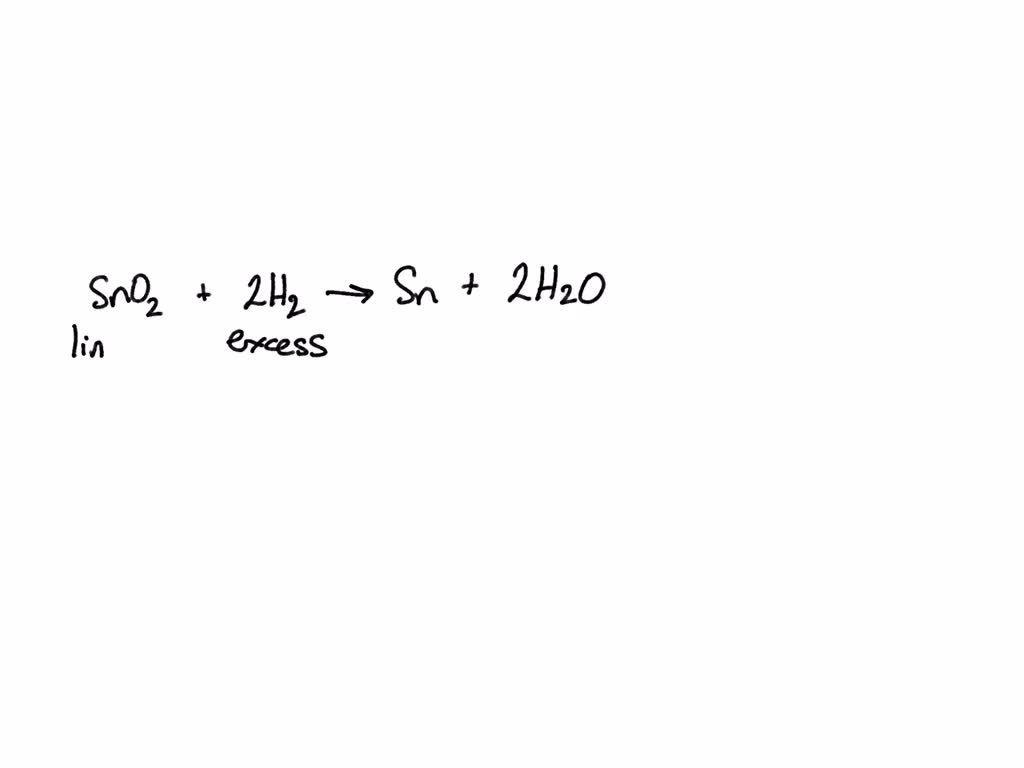
SOLVED: For the equation SnO2 + 2H2 Sn + 2H2O, tin (IV) oxide reacts with excess hydrogen to produce tin and water. What is the limiting reactant? A) SnO2 B) H2 C)

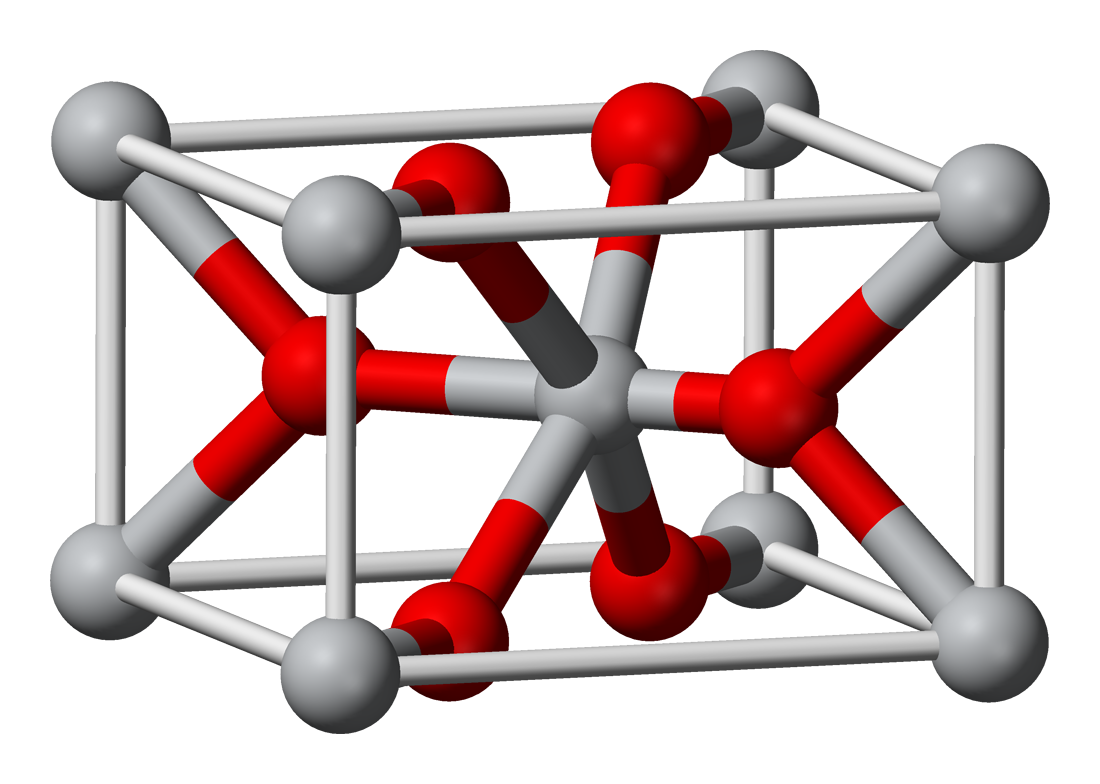
SOLVED:Smelting When tin(IV) oxide is heated with carbon in a process called smelting, the element tin can be extracted. SnO2(s)+2 C(s) →Sn(1)+2 CO(g) Interpret the chemical equation in terms of particles, moles,

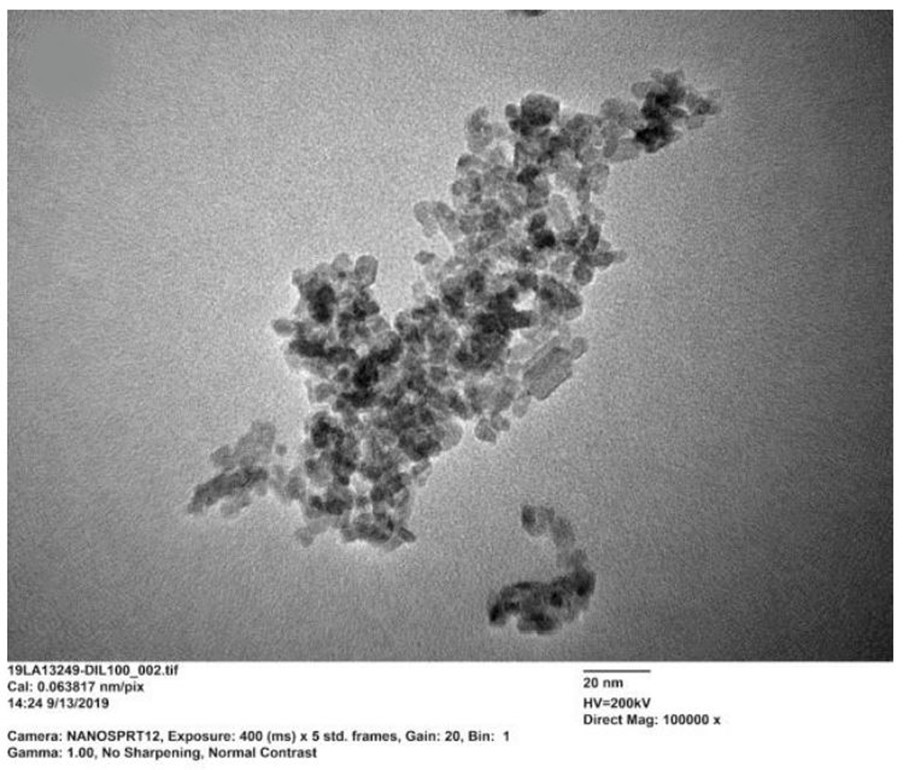
Resistance of the synthesized tin (IV) oxide samples 0DSnO2 (curves 1,... | Download Scientific Diagram

Write the formula of the following compunds: (i)Tin (IV) oxide ,(ii) Copper (I) iodide ,(iii) chromium (III) oxide ,(IV) Thallium (I) suplhate ,(V) Bismuth (III) niitrate Mercury (II) chloride

Resistance of the synthesized tin (IV) oxide samples 0DSnO2 (curves 1,... | Download Scientific Diagram

Tin (iv) oxide of 1,0 kg of those 22516-77 h in Uralsk online-store Topan, TOO | Buy Tin (IV) oxide of 1,0 kg of THOSE 22516-77 h Uralsk (Kazakhstan) | Topan, TOO : Allbiz